|
12/12/2023 0 Comments Serial dilution calculator 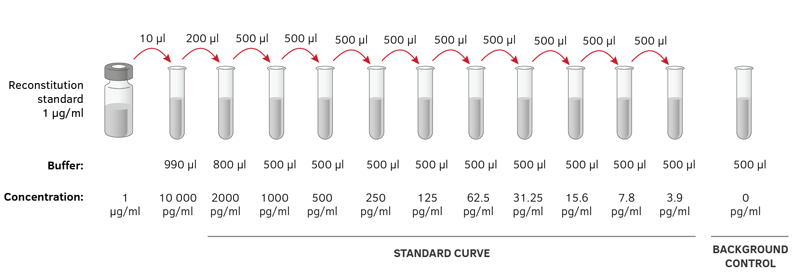
But if you want to perform the calculation by hand, use the following formula: When using an online calculator, input these values in the required fields to get the dilution factor along with the dilution performed in relative notation. In such a case, the value of the final volume is the original volume plus the value of the added volume. You can also get the volume you’ve added to the solution to dilute it. How do you calculate dilution factor?īefore calculating the dilution factor, you need two values namely the original volume of the diluted solution and the final volume of the solution after dilution. Although medication dilution is also applicable for pediatric practice, there are other factors you must also consider. This is important because dosages must be very accurate before they’re administered to infants and young children. In pediatric practice, professionals calculate medication dosages based on the weight of the child. Also, those who will compound or dilute medications must use the correct algebra formula if when performing the computations by hand. While you can also use this solution dilution calculator for medication dilution, you must know more about the medication you plan to dilute before administering it to a patient. The formulas which are typically used for this process are only useful for diluting medications from a higher concentration percentage to a lower one. 
Medication dilution is a lot like dilution of a solution since it also refers to the process of decreasing a solution’s concentration when you add more solvent to it. 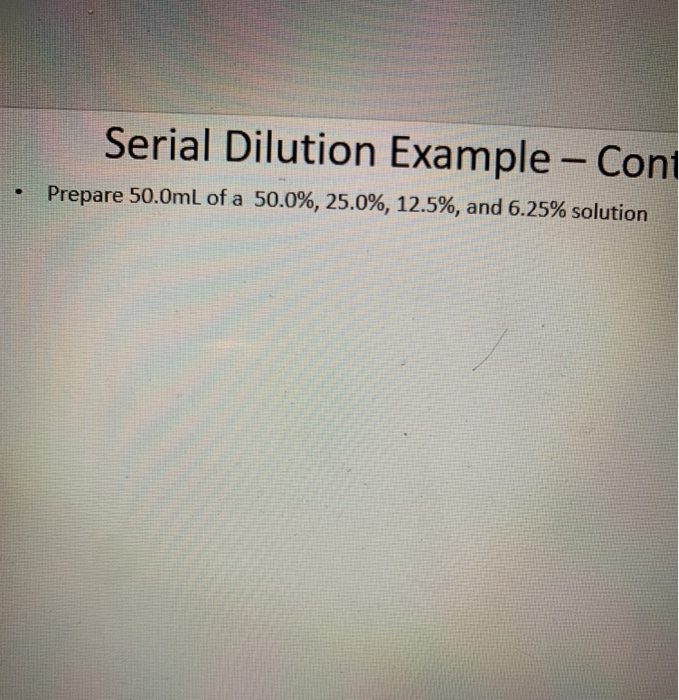
This results in a solution which is thoroughly mixed to ensure that all of the parts of your solution are identical. Therefore, to dilute concentration means that you add more solvent without adding more solute. You can do this by adding water to the solution or by adding more solvent to the solution. How do you dilute concentration?Īs aforementioned, the dilution of a solution refers to the process of reducing a solute’s concentration in a solution. However, it’s important to note that this formula isn’t equivalent to the proportion formula. V₂ refers to the volume of the diluted solution V₁ refers to the volume of the stock solution M₂ refers to the concentration of the diluted solution M₁ refers to the concentration of the stock solution Of course, you can also calculate this value by hand without using the molar dilution calculator. You can use this solution calculator to find out the amount you require if you would like to end up with a diluted solution of 200 mL that has a concentration of 20 mM. Let’s say, for instance, that you have a hydrochloric acid solution that’s very concentrated. The main purpose of this dilution calculator is to help you determine how to dilute a solution properly. To do this, you must mathematically calculate the volume of the solvent you would require to prepare the correct concentration of a new solution. In order for you to utilize a solution in a laboratory like for titration or for any other kind of process, you must accurately dilute it first to a lesser concentration. Diluting a solution is an important laboratory process since stock solutions are typically bought and stored in forms which are very concentrated. You can also dilute a solution by mixing a solution with a higher concentration with an identical solution with a lesser concentration. The process maintains the constancy of solute amount but it increases the solution’s total amount which, in turn, decreases the final concentration. Dilution of a solution refers to the process of adding a solvent to a solution for the purpose of decreasing the solution’s concentration. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
